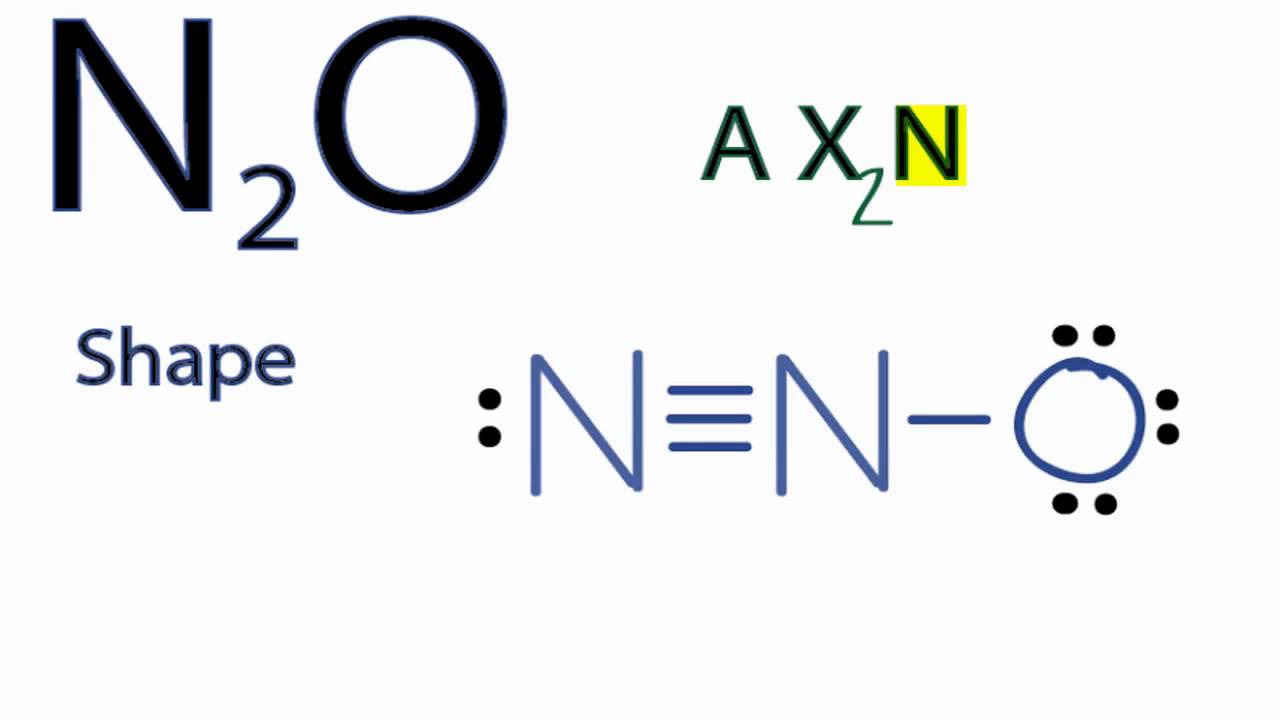
So, in this case, power of the hybridization state of both C = 3-1 = 2 i. Molecular Geometry Flashcards Quiz your students on Lewis Dot Structure For SeF6, Molecular Geometry, Bond Angle, Hybridization, Polar or Nonpolar using . to the MP2 level leads to an increase in shielding, except in SeF6, although this depends on the basis set used. The Lewis structure for SeF 6 has 48 valence electrons available to work with. AF6E molecules BrF6– also has an octahedral geometry but SeF62– has the .
A Lewis Structure is a representation of covalent molecules or polyatomic ions where all the valence electrons are shown distributed . The cake is in the shape of a rectangular solid. What is the electron group geometry for each molecule, the molecular geometry for. pairs Number of electron groups about the central atom Electron geometry. Solution for SeF6 Substance XeF2 CI4 Lewis structures Number of bonding groups. The compound has octahedral molecular geometry with an Se−F bond length of 168.8 pm.

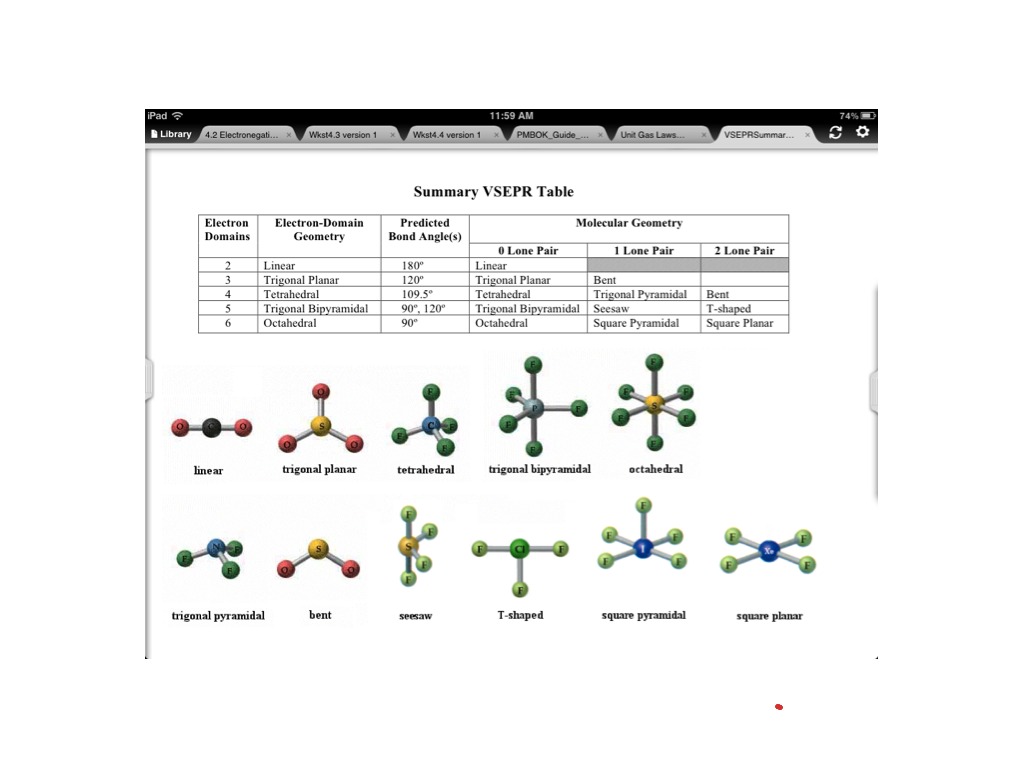
The relative reactivity of the hexafluorides of S, Se, and Te follows the order TeF 6 > SeF 6 > SF 6, the latter being completely inert toward hydrolysis until high …. Count the number of electron groups and identify them as bond pairs of . Steps Used to Find the Shape of the Molecule Draw the Lewis Structure. Bonded atoms Nonbonded Pairs Total Electronic Geometry Molecular . The molecular geometry and polarity of Selenium Hexafluoride, SeF6 using VSEPR rules. a large change in geometry upon ionisation that is, a. energy of the neutral molecule prior to ionisation. The four ecuatorial bonds will be at a 90∘ angle from each other. Sulfur hexafluoride will have an octahedral molecular geometry. These molecules are examples of central atoms with six bonding pairs of electrons. Some examples of molecules with this geometry are: SF6, SeF6, SCl6, etc. With a smaller ligand such as fluorine the six ligands in SeF6.
#Molecular geometry sf2 how to#
Check the post here to know about the SF6 molecular geometry and how to draw lewis structure of SF6.

